
U.S. quit smoking policy integrity drowns in pharmaceutical influence
Federal disclosure regulations designed to expose financial conflicts and protect the integrity and objectivity of taxpayer funded research were ignored by the Agency for Healthcare Research and Quality (AHRQ) when assembling panels to write U.S. smoking cessation policy in August 1998 and July 2006. Their discovery and application in 2007 revealed widespread financial conflicts which resulted in a damage control nightmare. The integrity of mandatory pharmacology use recommendations in both the June 2000 and May 2008 Clinical Practice Guideline for Treating Tobacco Use and Dependence are in question.
May 13, 2008 John R. Polito
On May 7, 2008 the U.S. government adopted an updated national tobacco dependency treatment policy, a policy that has sellers of FDA approved quit smoking products (Pfizer, GlaxoSmithKline and Johnson & Johnson) celebrating, while leaving those dedicated to non-pharmacology quitting shaking their heads in disbelief.
Written by a 24-member private sector panel of "experts" whose extensive pharmaceutical industry financial ties have the new policy mired in controversy, the 276 page updated "Guideline" again proclaims, that with few exceptions, all smokers trying to quit should be directed to go out and purchase pharmaceutical quitting products.
What should have been a rich and empowering tool to teach physicians critical lessons largely ignored by medical school educators (how to effectively counsel and support patient nicotine dependency recovery) the Guideline is instead little more than a pharmaceutical industry quitting product shopping guide.
While the May 2008 Guideline discloses a number of panel member pharmaceutical industry financial ties, the federal agency responsible for Guideline production and conflicts investigation, the AHRQ, substantially reduced the initial conflicts disclosure period, allowed panel members identified as having "significant financial interests" to remain on the panel, and in regard to the panel chairman, ignored substance in favor of form.
The disclosures fail to provide readers awareness of funds provided to panel members or their organizations by the Robert Wood Johnson Foundation (RWJF), an organization with strong ties to Johnson & Johnson, the company which likely holds the largest share of the U.S. nicotine replacement therapy (NRT) products market. But why care?
The Guideline panel summarily dismisses, as having "methodological constraints," a growing body of real-world evidence indicating that non-pharmacology quitting is superior. Why care? Because this national quitting method monopoly keeps quality non-pharmacology counseling and support oriented programs suppressed, hidden and forbidden as government referral resources. We should care because the panel gave Chantix an equal recommendation with NRT and Zyban when the FDA has linked Chantix to depressed mood, behavioral changes, suicidal thinking and suicide.
The June 2000 Guideline panel was the first to declare pharmacology use mandatory. It was authored by a panel on which 11 of 18 members openly acknowledged pharmaceutical industry financial ties. All eleven sought to join this panel, and last Wednesday all but one were identified as having again served as panel members.
Although survivors of eight years of government blacklisting for non-compliance, non-pharmacology quitting programs were provided a ray of hope by a February 8, 2007 front-page Wall Street Journal article by Pulitzer Prize winning journalist Kevin Helliker. Helliker questioned the policy and exposed financial conflicts.
The article should have caused the AHRQ to take swift action in fully investigating and removing all panel members having significant financial interests. It didn't. After reading it, one would think it would have caused panel members suffering from the "Dr. Drug Rep" syndrome to awaken and act responsibly. It didn't. Instead, those having significant financial and conflicts of interests, as defined by federal law, decided to stay.
In November 2007, the U.S. Centers for Disease Control announced that decline in the U.S. smoking rate had ground to a standstill. It seemed unthinkable that government health officials would continue to ignore the fact that since the June 2000 Guideline had effectively declared our nation's most productive quitting method in violation of U.S. quitting policy - non-pharmacology cold turkey quitting - that national cessation has floundered horribly.
The financial ethics nightmare infecting AHRQ Guidelines is of national concern. Smoking is our nation's leading cause of disease and pre-mature death. Through Medicare, Medicaid and higher hospital costs due to treating uninsured smoker diseases, we are each stakeholders. We've watched as the pharmaceutical empire has increasingly convinced health bureaucrats to use precious tax dollars to pay for free nicotine replacement products, products that when used as stand-alone quitting aids generate dismal 7% six-month quitting rates.
Health professors question May 2008 Guideline update
"Far from being an objective review and assessment of the best possible strategy to enhance smoking cessation among smokers in the United States," wrote Dr. Michael Siegel, a physician and professor at Boston University School of Public Health, "the guideline is a heavily biased analysis that is plagued by the presence of severe financial conflicts of interests among the panel's chair and at least 8 of its other members."
According to May 2008 Guideline Appendix A "Financial Disclosures" (starting on PDF page 223), 3 of 28 panel members had financial interest conflicts so serious that they were excused from the panel after substantial service, possibly more than a year. One additional member voluntarily resigned. Three others were determined to have significant financial interests of at least $10,000 but for unstated reasons were allowed to remain. Six others were found to have financial interests that were not significant (less than $10,000).
What the May 2008 Guideline disclosures fail to indicate is that at least 2 panel members dismissed for serious conflicts were allowed to author the June 2000 Guideline, the Guideline panel which first took the drastic step of making pharmacology use recommendations mandatory throughout government. This effectively destroyed program credibility of thousands of neighborhood based quitting programs grounded in counseling and ongoing support.
"What this means is that we basically have a group of experts who are largely financially linked with pharmaceutical companies, and who stand to gain personally if they recommend the use of pharmaceuticals as part of their smoking cessation guidelines," wrote Dr. Siegel. "The more use of pharmaceuticals they recommend, the more they stand to gain."
"This is a conflict of interest in the ugliest way that I can imagine. It is precisely the type of thing that needs to be stopped," Siegel wrote.
"Ironically," says Dr. Siegel, "it is the Department of Health and Human Services, the Public Health Service, and NIH itself which are regulating conflicts of interest among America's medical researchers. To have such a huge conflict of interest in its own supported work is unconscionable."
"While I find it objectionable that these agencies would allow experts with these type of financial conflicts of interest to serve on the panel, I find it equally troubling that such individuals would agree to serve. I believe that based on these conflicts, these experts should have recused themselves from service on the panel."
As Dr. Siegel notes, "The conflict in this case is not just a hypothetical one. It reveals itself in the extreme bias of the guideline. The analysis over-estimates the benefit of drugs in smoking cessation and overlooks population-based evidence showing that most people who quit smoking do so without pharmaceutical aids."
A May 8 Associated Press story quotes Dr. Lois Biener, a researcher at the University of Massachusetts Boston, as agreeing with Dr. Siegel. "There's little evidence that these drugs are superior in the long run to quitting without help, and while a few studies have shown some benefit, it's 'way less than what is claimed' by medication advocates."
"Most people who quit do so without smoking-cessation drugs, said Dr. Biener."
Regarding Chantix, equally disturbing is why 24 panel members, who within the Guideline refer to themselves eight times as "experts," would within those 276 pages have absolutely no risk-benefit discussion about recommending Chantix, which has quickly and clearly become the most dangerous quitting product ever.
Imagine "experts" writing a "guideline" that fails to guide health care providers in helping patients decide whether risking their life by using a new quitting product is worth it. Imagine a Guideline that recommends use of Chantix (varenicline) on PDF pages 5, 7, 25, 60 and 62 but waits until page 63 to first mention its association with suicide.
If Dr. Siegel and Dr. Biener are upset now, their concerns will likely grow once they more closely scrutinize pharmaceutical influence at play among panel members.
Freedom of Information Act used to obtain panel information
The day after Helliker's February 8, 2007 story, I e-mailed Dr. Carolyn Clancy, Director of the Agency for Healthcare Research and Quality (AHRQ) and pursuant to the Freedom of Information Act (FOIA) requested "the names of all panel members, their bios and their smoking cessation product conflicts disclosures, including how much they have accepted, when, the reason/s for payment, and from which pharmaceutical interests, or point me to Internet links disclosing this extremely critical public information."
On March 21, 2007, I was sent 825 pages of panel member resumes, what academics refer to as "curriculum vitaes" or CVs. Included were CVs of two panel members who, on some unknown date after March 21, 2007, no longer served on the panel.
On September 5, 2007, I was sent 24 signed "Panel Agreement and Financial Disclosure" forms. Nineteen were signed during July 2006, three were signed or dated in August, and the last two, those of 1996, 2000 and 2008 Guideline panel Chairman Michael Fiore and Michael Goldstein, bore the same date, 9/5/06. Each disclosure form requested "complete" disclosure of all potential financial conflicts for the "past 5 years." I could make out the signatures of all panel members except three, and each of those denied any financial interest.
The May 2008 Guideline discusses the financial disclosure process at PDF page 223. It asserts that, "Prior to the second in-person Panel meeting in June 2007, and before any decisions regarding Panel recommendations were made, Panel members were required to complete a more exhaustive disclosure process for calendar years 2005, 2006, and 2007, based on the United States Department of Health and Human Services, PHS Title 42, Chapter 1, Part 50 guidelines for the conduct of research (http://ori.hhs.gov/fedreg42cfr50)."
The above federal regulation is not simply some "guideline" but the "law of the land" and mandatory. Section 42 CFR 50.604(g)(2) required the reporting of any conflicting interest prior to expenditure of any funds, not a full year into the panel's work.
The above Guideline statement has been modified from the one appearing in the October 2007 draft which asked readers to believe that this had been a planned process. The draft asserts, "This evaluation process entailed a rigorous and transparent two-step procedure for disclosing and handling conflict of interest on the part of panel members."
How transparent is a process when those submitting FOIA disclosure requests are not sent the more "exhaustive" disclosures? I was sent 2006 disclosures in September 2007.
Fiore's website time-line asserts that the decision to update the Guideline occurred on July 1, 2006 (50 days after the FDA announced Chantix approval), which explains why nearly all the financial disclosures I received were signed in July 2006, eleven by July 8. But how is it "more exhaustive" to narrow the pre-Panel examination window from the five year disclosures I was provided, to just 18 months prior to selecting panel members?
Did AHRQ use science integrity regulations to prevent transparency?
Did AHRQ administrators intentionally narrow both the conflicts time window and its scope so as to avoid labeling the Panel's chairman, Fiore, as having a "significant financial interest"? Guideline page 223 asserts that a financial conflict is deemed significant if "net reportable compensation in excess of $10,000 in any reporting year."
In May 2005, Fiore testified (see PDF page 14) that, "[o]ver the past five years, my outside consulting work on an annual basis has ranged between about $10,000 and $30,000 or $40,000 per year."
No language in federal disclosure law told AHRQ officials to limit investigation for significant financial interests to 18 months instead of 3 or 5 years.
42 CFR 50.601 tells the AHRQ that these regulations are intended as a flexible common sense tool to promote "objectivity in research by establishing standards to ensure there is no reasonable expectation that the design, conduct, or reporting of research funded under PHS grants or cooperative agreements will be biased by any conflicting financial interest of an Investigator."
What are "we the people" to think when our own government, the Agency for Healthcare Research and Quality, the federal gatekeeper agency charged with guarding research integrity, appears to have used financial conflicts laws as shields to knowingly hide serious investigator financial conflicts from public view?
Fiore also testified in May 2005 as to receiving an annual $50,000 per year unrestricted chair endowment grant from funds given to the University of Wisconsin by GlaxoSmithKline (then Glaxo Wellcome). "GlaxoSmithKline gave a grant to the University of Wisconsin that established a chair for the treatment of tobacco dependence," Fiore testified. "That donation by GlaxoSmithKline was to the University. Named chairs at the University of Wisconsin provide the person who sits in that chair to access to the revenue generated from the investment on the initial grant. So in this instance, I have access to up to $50,000 per year to support my University approved and sanctioned educational, research, and policy activities."
Resolution 7594 in the minutes of the December 5, 1997 meeting of the University of Wisconsin Board of Regents (see PDF page 15) appears to support Fiore's contention that the University not Glaxo Wellcome made the decision to honor Fiore with this professorship chair. "That, upon recommendation of the Chancellor of the University of Wisconsin-Madison and the President of the University of Wisconsin System, Professor Michael C. Fiore, Department of Medicine, be appointed to the Glaxo Wellcome Professorship for Study of Tobacco Dependence, effective immediately."
On May 10, 2008 I e-mailed the University of Wisconsin Foundation asking "whether Glaxo Wellcome or the University of Wisconsin selected Dr. Fiore as the original endowment chair recipient." The Foundation's Vice-President of Legal Affairs responded on May 12 indicating, "I can tell you that the agreement mentions no individual by name. It is intended to support the University's Center for Tobacco Research and Intervention and supports the Director of that Center."
During a follow-up call, it was made clear that Glaxo Wellcome directed that the funds be used to support the Director of the Center for Tobacco Research and Intervention, who was then Dr. Michael Fiore.
If no exemption applies, Glaxo Wellcome's $50,000 per year gift to Fiore is five times greater than necessary to declare a "significant financial interest" under 42 CFR 50.603.
Can federal regulations intended to protect scientific integrity from financial influence be bypassed by use of university professorship chair endowments? If so, what government produced science relying upon professors effectively placed on the industry's permanent payroll will be worthy of belief?
Fiore's May 2005 testimony also suggested that the University of Wisconsin Center for Tobacco Research and Intervention (UW CTRI) receives in the neighborhood of $1 million per year to conduct research for pharmaceutical companies. In that the May 2008 Guideline declares AHRQ to have developed the Guideline in "partnership" with both the UW CTRI and Robert Wood Johnson Foundation (RWJF), and federal disclosure regulations require disclosure by public or private "Institutions" as well as "Investigators," where are the UW CTRI's and RWJF's conflicts disclosure?
After Helliker's story, did AHRQ look back to see if the integrity of the 1996 and 2000 Guidelines were compromised by significant conflicts of interest? On December 12, 1996 Fiore declared at an FDA advisory committee meeting that I "am speaking today as a consultant to Glaxo Wellcome." He states, "I served ... as Chair for the Clinical Practice Guideline on Smoking Cessation," released in April 1996.
Fiore research funded by Glaxo Wellcome was published in March 2000, the same period during which he chaired the June 2000 Guideline. A 2002 study financial disclosure in the journal Chest asserts, "Dr. Fiore has served as a consultant for, given lectures sponsored by, or has conducted research sponsored by Ciba-Geigy, SmithKline Beecham, Lederle Laboratories, McNeil Consumer Products, Elan Pharmaceutical, Pharmacia, and Glaxo Wellcome."
Why were Robert Wood Johnson Foundation relationships ignored?
The Robert Woods Johnson Foundation's (RWJF) hands are all over the U.S. Guidelines. Its hands are also all over Guideline panel members. Not only did RWJF partner in funding both the June 2000 and May 2008 Guidelines, it approved and awarded Guideline panel members research project funding while in service as panel members.
Robert Wood Johnson II built the original 1886 three brother family business of Johnson & Johnson (J&J) into the world's largest health products company. Today the Johnson & Johnson "Family of Companies," subsidiaries with interlocking management, number 250. They include the ALZA Corporation which created the NicoDerm® CQ® and Clear NicoDerm® CQ® nicotine patches, McNeil Consumer Healthcare which manufactures the Nicotrol nicotine patch (that on July 3, 1996 became the first FDA patch approved for over-the-counter sales), licenses sales of Nicoderm nicotine patches and Nicorette® nicotine gum, and markets the Nicotrol inhaler, and McNeil Pharmaceuticals which the FDA authorized to market Nicotrol nasal spray on July 3, 1996.
When Robert Wood Johnson II died in 1968 his will devised his 10,204,377 shares of Johnson & Johnson common stock to establishment of a foundation, the foundation that today bears his name, The Robert Wood Johnson Foundation (RWJF). As of 2006, RWJF owned 55,983,308 shares of J&J common stock valued at $33.4 billion dollars.
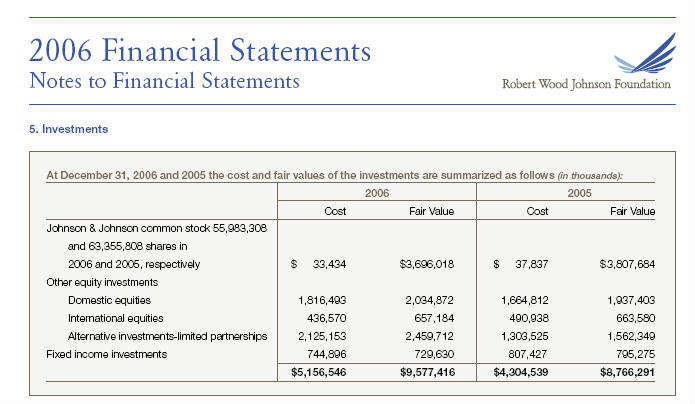
In June 2006, J&J announced it was buying Pfizer's Consumer Healthcare business for $16.6 billion dollars. The purchase was concluded on December 20, 2006 with J&J announcing that with "the acquisition, the Johnson & Johnson Consumer Group portfolio will now feature products such as ... the NICORETTE line of smoking cessation treatments."
Since its creation, the RWJF Board of Trustees charged with governing the foundation has included many J&J executives and one namesake. Today RWJF's website reports that Trustee Robert E. Campbell is a retired vice chairman of the board of directors of J&J, Trustee Edward J. Hartnett is a retired company group chairman of Johnson & Johnson, grandson and heir Robert Wood Johnson IV is a Trustee, Trustee George S. Frazza "previously served Johnson & Johnson for more than 30 years in the roles of corporate secretary, vice president and general counsel, and was a member of the executive committee," and Trustee Ralph S. Larsen is a former chairman of the board and CEO of Johnson & Johnson.
According to RWJF, it contributed $102,016 to the University of Wisconsin School of Medicine with Michael Fiore as overseer, toward creation of the June 2000 Guideline. "On September 14-15, 1999, panel members met in Madison, Wis., where they evaluated their findings and prepared a draft of the new Guideline," asserts the RWJF. "The final version was sent to RWJF, and submitted to the USPHS for approval."
On December 20, 2007, the RWJF disclosed it had awarded the University of Wisconsin School of Medicine and Public Health $100,000 for "updating, testing and disseminating the 2008 tobacco treatment guideline," with Fiore listed as one of two project directors.
The RWJF's primary asset is J&J stock. Its primary income is by earnings from, and sales of, J&J common stock. RWJF's financial well being is tied directly to J&J's stock values. Would the RWJF foundation dare make decisions harmful to its own economic interests? Has the RWJF ever once reached out to the 80-90% of smokers annually voting to attempt quitting without medications, and attempted to help save their lives by creating access to high quality non-pharmacology counseling and support programs that it knows would dramatically increase their odds of success?
Is it realistic to expect the RWJF to ever fund research that might tend to prove that Johnson & Johnson quit smoking product efficacy findings are grounded in sham clinical trials that were never blind as claimed? Does the RWJF Board of Trustees truly believe that researchers discovered some magic means to hide from nicotine addicts volunteering to participate in "medicine" clinical trials, the fact that they had instead been randomly assigned to receive a placebo device, and were, contrary to their expectations, now experiencing the onset of full-blown nicotine withdrawal? Is RWJF's focus on the integrity and quality of medical science or, in this case, its use to bolster sales and stock values?
If current federal financial disclosure regulations exempt nearly "anything of monetary value" flowing to an "Investigator" from non-profit entities ( 42 CFR 50.601), should they? Should a paid employee of the RWJF have been allowed to sit on a panel authoring U.S. cessation policy? Would all pharmaceutical companies be wise to establish their own foundation, allowing them not only to shield and hide researcher financial ties but to participate in writing government policy favorable to their founding corporation's economic interests?
Panel member financial disclosures and relationships
NOTE: In determining existence of a "significant financial interest" as they relate to panel member RWJF relationships listed below, current federal disclosure regulations exempt "all income from seminars, lectures, or teaching engagements sponsored by public or nonprofit entities," and "income from service on advisory committees or review panels for public or nonprofit entities."
The below panel members either made a financial disclosure that appeared within the June 2000 Guideline, the May 2008 Guideline, made an industry link disclosure elsewhere, or had some prior relationship with the Robert Wood Johnson Foundation. The CVs of three panel members, although expert in other fields, evidenced little or no experience in the field of smoking cessation.
- Bailey's May 2008 Guideline Disclosure: "William C. Bailey reported significant financial interests in the form of compensation from three different pharmaceutical companies in 2006 and two in 2007 for speaking engagements."
Bailey's five year Financial Disclosure dated 7/6/06 states in part, "I, William C. Bailey, have received research contracts from drug companies as sponsors of investigational drug research projects; I receive honoraria from pharmaceutical companies for delivering lectures as part of Continuing Medical Education programs for physicians with appropriate disclosure; ... Some of the pharmaceutical companies are: GlaxoSmithKline ... Pfizer ... "
Bailey's June 2000 Guideline Panel Member Financial Disclosure states, "William C. Bailey has served as a consultant for, given lectures sponsored by, or has conducted research sponsored by Ciba-Geigy, SmithKline Beecham, Lederle Labratories, NcNeil, Elan Pharmaceuticals, and Glaxo Wellcome.
- Baker's May 2008 Guideline Disclosure: "Timothy B. Baker reported no significant financial interests. Under additional disclosures, he reported that he has served as a co-investigator on research studies at the University of Wisconsin that were sponsored by four pharmaceutical companies."
Baker's July 3, 2006 five year Financial Disclosure in part asserts that three of those companies were "Pfizer, Nabi and Glaxo Wellcome."
Baker's CV indicates that he holds the position of "Director of Research, UW Medical School - Center for Tobacco Research and Intervention (UW-CTRI)." [note: UW-CTRI was founded in 1992 by Michael Fiore, where today Fiore serves as Director and Baker serves as Associated Director]
- Benowitz's May 2008 Guideline Disclosure: "Neal L. Benowitz reported significant financial interest in the form of compensation from one pharmaceutical company for each of the years 2005-2007, as well as stock ownership in one pharmaceutical company. Under additional disclosures, he reported providing expert testimony in lawsuits against tobacco companies."
Benowitz's five year "Financial Disclosure dated 8/24/06 in part asserts, "Pfizer - scientific advisory board, GlaxoSmithKline - occasional board consultant, Nabi - occasional paid consultant .... Stock ownership Pfizer, Alexza ..."
Benowitz's CV indicates service from "2003-now Robert Wood Johnson Foundation, Smoking Cessation Leadership Center."
- Curry's May 2008 Guideline Disclosure: "Susan J. Curry reported no significant financial interests and no additional disclosures."
Curry's 7/12/06 five year Financial Disclosure states, "Paid honoraria for scientific consultation by Pfizer, Inc, Sanofi-Aventis
Curry's CV indicates that from 2/04 to Present she has served on the Board of Directors for the American Legacy Foundation (ALF) [Note, fellow 2008 Guideline panel member Cheryl Healton serves as ALF President and CEO. Also of interest is that Dr. Steven A. Schroeder served as CEO and President of the Robert Wood Johnson Foundation from 1990-2002. An ALF board member since 2000, in Feb. 2003 Dr. Schroeder was named Chairman of the ALF Board of Directors, a position he held until November 2005. On January 3, 2007, during the Guideline update process, RWJF awarded ALF a grant of $720,000 to "help American adults stop smoking" using "evidence based resources."
RWJF awarded a grant of $2,996,867 on 01/25/05 and apparently another of $1,240,783 on 04/23/07 under which Curry is project director.
RWJF also lists Curry & Fiore as Program Directors and Orleans (as RWJF staff) in running its "Addressing Tobacco in Healthcare" website.
- Dorfman's May 2008 Guideline Disclosure: "Sally Faith Dorfman reported no significant financial interests. Under additional disclosures, she reported her employment by Ferring Pharmaceuticals, Inc., a company whose business does not relate to treating tobacco dependence."
Dorfman's 7/10/06 five year Financial Disclosure declares "I have no relevant financial interest in the guideline panel or resulting guideline."
Dorfman's Appendix C June 2000 Guideline Panel Member Financial Disclosure states, "Sally Faith Dorfman has served as a consultant for, given lectures sponsored by, or conducted research sponsored by various pharmaceutical companies."
Dorfman's CV indicates that her current position is as "Associate Director, Medical Affairs (Infertility), Ferring Pharmaceuticals, Suffern, New York" since 2006. She served on the Robert Wood Johnson Foundation National Advisory Committee, Smoke-Free Families from 1995-1999.
- Fiore's May 2008 Guideline Disclosure: "Michael C. Fiore reported no significant financial interests. Under additional disclosures, he reported that he served as an investigator on research studies at the University of Wisconsin (UW) that were supported wholly or in part by four pharmaceutical companies, and in 2005 received compensation from one pharmaceutical company. In addition, he reported that, in 1998, the UW appointed him to a named Chair, which was made possible by an unrestricted gift to the UW from GlaxoWellcome."
Fiore's Panel Agreement and Financial Disclosure dated 09/05/06 advises him that it "applies to the past 5 years" and that he disclose completely all affiliations with or financial involvement, including employment, consultancies, honoraria, stock ownership or options or grants with any entity having a financial interest in or financial conflict with guideline matter or materials. Fiore chose to attach a typed, signed document he entitled "Statement of Disclosure" in which he asserts, "In keeping with recent JAMA recommendations, Dr. Fiore does not accept honorarium nor do consulting work for the pharmaceutical industry. In 1998, the University of Wisconsin (UW) appointed Dr. Fiore to a named Chair, made possible by an unrestricted gift to UW from Glaxo Wellcome."
As indicated above, Glaxo Wellcome directed the University of Wisconsin that the only person who could sit in the $50,000 per year professorship chair was the Director of Center for Tobacco Research and Intervention, who Glaxo Wellcome knew to be Dr. Fiore.
In a February 7, 2007 WSJ article Fiore claimed to have stopped accepting pharmaceutical industry quit smoking product consulting fees and honorariums in 2005.
Fiore's testimony on or about May 9, 2005 in U.S. v. Philip Morris does not mention that he stopped accepting consulting fees. He testifies on PDF page 14 that, "Over the past five years, my outside consulting work on an annual basis has ranged between about $10,000 and $30,000 or $40,000 per year." He testifies on page 15 that each year he receives another $50,000 from the Glaxo Wellcome Chair he occupies and references above.
The website of the University of Wisconsin Center for Tobacco Research and Intervention (UW-CTRI), founded by Fiore in 1992, and which he continues to direct, does not provide visitors with any statement as to financial conflicts.
A May 8, 2008 article by Michael Siegel, MD of Boston University School of Public Health challenged Fiore's ethics for failure to disclose potential financial conflicts of interest in a just released article Fiore co-authored, which appeared in the American Journal of Public Health. In the article Fiore advocated increased access to pharmaceutical treatment for smoking cessation.
Fiore's Appendix C June 2000 Guideline Panel Member Financial Disclosure stated, "Michael C. Fiore has served as a consultant for, given lectures sponsored by, or has conducted research sponsored by Cib-Geigy, SmithKlineBeecham, Lederle Labratories, McNeil, Elan Pharmaceutical and Glaxo Wellcome."
Fiore's CV indicates that from "2006-Present" he has served as "DIRECTOR, Robert Wood Johnson Foundation initiative - Addressing Tobacco in Healthcare. It further indicates that from "1997-2006" Fiore served as "DIRECTOR, Robert Woods Johnson Foundation National Program Office - Addressing Tobacco in Managed Care."
On 12/19/03 RWJF awarded UW-CTRI, with Fiore as project director, $300,000 to translate tobacco dependence research into policy; on 01/17/05 $400,000 to evaluate the national quit line, on 07/27/05 $400,000 to build a network to fund tobacco related health care research; on 09/24/07 $99,316 to increase Medicaid recipient demand for tobacco dependence treatment; and, most recently, on 12/20/07 $100,000 for "Updating, testing and disseminating the 2008 tobacco treatment guideline."
RWJF also lists Fiore and Curry as Program Directors and Orleans (as RWJF staff) in running its "Addressing Tobacco in Healthcare" website.
Although unknown whether Fiore was compensated or any compensation's source, Fiore was moderator of a 09/18/07 Wisconsin "Dialogue" on smoking's economic impact, as part of a program listing Pfizer as one its "Benefactors."
- Froehlicher's May 2008 Guideline Disclosure: "Erika S. Froehlicher reported no significant financial interests and no additional disclosures."
Froelicher's CV indicates that, as with fellow panel member Benowitz, she is a professor at UCSF, in 2002 they co-authored an article entitled "Women's Initiative for Nonsmoking: Under-use of nicotine replacement therapy (NRT)," and a second article in 2003. In August 2001 Froelicher was an invited lecturer for RWJF on smoking in nurses and during two days in August, 2003 served as a reviewer for the RWJF.
- Goldstein's May 2008 Guideline Disclosure: "Michael G. Goldstein reported no significant financial interests. Under additional disclosures, he reported that his employer received support from Bayer Pharmaceutical prior to 2005 and that he was employed by Bayer Pharmaceutical Corporation prior to January 1, 2005. His organization received payments for his professional
services from two pharmaceutical companies and one commercial Internet smoking cessation site during the period 2005-2007."
Michael G. Goldstein's 9/5/06 five-year Financial Disclosure forwarded to me by the DHHS under cover letter of 09/05/07 states in part, "Bayer Pharmaceutical Corporation - employee 9/98-12/04 (however Bayer produces no tobacco dependence products) .... Pfizer - have served on advisory panels for Health Education Principles - remuneration received by Institute for Healthcare Communication."
Goldstein's Appendix C June 2000 Guideline Panel Member Financial Disclosure stated, "... in addition to being an employee of the Bayer Corporation, has served as a consultant for, given lectures sponsored by, or had conducted research sponsored by Glaxo Wellcome, McNeil, Ciba-Geigy, SmithKline Beecham ..."
- Healton's May 2008 Guideline Disclosure: "Cheryl Healton reported no significant financial interests and no additional disclosures."
As indicated above, Healton is CEO of American Legacy Foundation (ALF). On January 3, 2007, during the Guideline update process, RWJF awarded ALF a grant of $720,000 to "help American adults stop smoking" using "evidence based resources."
- Henderson's May 2008 Guideline Disclosure: "Patricia Nez Henderson reported no significant financial interests and no additional disclosures."
On 12/07/07 a $49,969 grant by RWJF was awarded to develop tobacco-free and wellness policies for the Navajo nation with Henderson as project director.
- Jaén's May 2008 Guideline Disclosure: "Jaén Carlos Roberto Jaén reported no significant financial interests and no additional disclosures."
It would appear that Jaén was named "Panel Vice Chair" to deflect fall-out from the 02/08/07 Wall Street Journal article. Jaén's study history indicates that in 1994 he co-authored a primary care nicotine patch study and in 1997 was lead author in a Medicaid study involving free nicotine patches.
Jaén's CV indicates that in 1994 he was a RWJF Generalist Physician Faculty Scholar. Jaén's biowebpage asserts he is a "Member of the National Advisory Committee for Robert Wood Johnson Foundation's 'Prescribe for Health Program.'"
Jaén's Appendix C June 2000 Guideline Panel Member Financial Disclosure stated he "has served as a consultant for, given lectures sponsored by, or has conducted research sponsored by Glaxo Wellcome Pharmaceuticals."
- Kottke's May 2008 Guideline Disclosure: "Thomas E. Kottke reported no significant financial interests and no additional disclosures."
Kottke's July 6, 2006 signed Guideline Panel Financial Disclosure asserts, "I have no relevant financial interest in the guideline panel or resulting guideline."
A June 2007 paper co-authored by Kottke, entitled Lipid Management in Adults, contains a conflicts disclosure stating, "Thomas E. Kottke, MD has received speaker fees or honorariums from ASTRA-Zeneca, Pfizer and Sanofi-Aventis.
Kottke's Appendix C June 2000 Guideline Panel Member Financial Disclosure asserted that he, "has served as a consultant for, given lectures sponsored by, or has conducted research sponsored by McNeil Consumer Healthcare." McNeil is a division of Johnson & Johnson which sells Nicorette gum.
Kottke's bio webpage indicates he was a RWJF Clinical Scholar.
Interestingly, Kottke wrote a 01/18/95 editorial entitled "Nicotine Patches: a Doubtful Key to Tobacco Control?"
- Lando's May 2008 Guideline Disclosure: "Harry A. Lando reported no significant financial interests. Under additional disclosures, he reported serving on an advisory panel for a new tobacco use cessation medication and attending 2-day meetings in 2005 and 2006 as a member of this panel."
Lando's online "Vita" dated January 2008 asserts on pages 4-5 that he is a consultant to Pfizer Pharmaceuticals, Robert Wood Johnson Foundation and SmithKline Beecham.
Lando's Appendix C June 2000 Guideline Panel Member Financial Disclosure stated he, "has served as a consultant for, given lectures sponsored by, or has conducted research sponsored by Glaxo Wellcome and SmithKline Beecham."
As fund raising chairman for the 2008 World Conference on Tobacco or Health to be held in India, Lando arranged for Pfizer and GlaxoSmithKline to serve as conference sponsors.
Lando was quoted in Helliker's 02/08/07 Wall Street Journal article as saying, "I view the pharmaceutical industry as our ally."
- Mecklenburg's May 2008 Guideline Disclosure: Robert E. Mecklenburg reported no significant financial interests. Under additional
disclosures, he reported assisting Clinical Tools, Inc., through a governmental contract to develop a PHS 2000 Guideline-based Internet continuing education course.
Mecklenburg's Appendix C June 2000 Guideline Panel Member Financial Disclosure stated he, "has served as a consultant for, given lectures sponsored by, or has conducted research sponsored by SmithKline Beecham and Glaxo Wellcome.
Mecklenburg's CV appears dated but does assert "Contracts of varying length have been held between 1988 to 2006." "3. Pharmaceutical companies:" Glaxo Wellcome SmithKline Beecham."
- . Mermelstein's May 2008 Guideline Disclosure: Robin Mermelstein reported no significant financial interests and no additional disclosures.
Mermelstein's CV asserts,"1997-2005 Robert Wood Johnson Foundation Scientific Advisory Committee Member: Addressing Tobacco in Managed Care." Also, "1999-2005 Robert Wood Johnson Foundation Tobacco Etiology Research Network, Committees on Adolescent Measures of Use and Measures of Dependence; Context Committee. Also, "2000-2005 Member, Expert Advisory Panel, Robert Wood Johnson Foundation" youth use & cessation survey.
- Mullen's May 2008 Guideline Disclosure: Patricia Dolan Mullen reported no significant financial interests and no additional disclosures.
Mullen's CV dated 08/06 CV indicates, "Robert Woods Johnson Foundation: National Advisory Committee, Smoke-Free Families (2000-2006)
- Orleans' May 2008 Guideline Disclosure: C. Tracy Orleans reported significant financial interests in the form of a dependent child who owns pharmaceutical stock, and no additional disclosures. Lawrence Robinson reported no significant financial interests and no additional disclosures.
Orleans' July 26, 2006 signed Financial Disclosure states, "I have no relevant financial interests in the guideline panel or resulting guideline."
RWJF claims Orleans is the RWJF's senior scientist and "leads the Foundation's efforts to develop and disseminate science-based strategies for addressing the major behavioral causes of preventable death and chronic disease" "She oversees a current portfolio of more than $375 million in RWJF national programs and grants." "Recruited to the Foundation in 1996 as a national leader in tobacco control and health behavior change research and practice, Orleans focused through 1999 on expanding the Foundation's investments in policy-based approaches to tobacco cessation ..." "She is especially pleased that her early innovative work in tobacco control and cessation programs continue to have wide impact in helping smokers quit, including the Johnson & Johnson worksite tobacco control program; 'Free & Clear'..."
RWJF identifies Orleans as "RWJF staff" in running its "Addressing Tobacco in Healthcare" website, with Fiore and Curry as Directors.
Orleans and Fiore co-authored papers in 2002 (also with Curry), 2004, 2005, 2006 and 2008.
- Stitzer's May 2008 Guideline Disclosure: Maxine L. Stitzer reported no significant financial interests. Under additional disclosures, she reported participation on a pharmaceutical scientific advisory panel for a new tobacco use cessation medication.
Stitzer's signed 7/5/06 five-year Financial Disclosure does not reference the above but instead states, "Pfizer Pharmaceuticals - promotional speaker."
Footnotes to a November 2006 study recites that "Maxine L. Stitzer, Ph.D., has received promotional speaker support from Pfizer."
Stitzer's Appendix C June 2000 Guideline Panel Member Financial Disclosure stated she, "has served as a consultant for, given lectures sponsored by, or has conducted research sponsored by McNeil and SmithKline Beecham.
Stitzer's CV asserts that since 2000 she has been on an advisory board to "University of Wisconsin Center for Tobacco Research & Intervention" a/k/a Fiore's UW-CTRI
- Wewers's May 2008 Guideline Disclosure: Mary Ellen Wewers reported no significant financial interests and no additional disclosures.
Wewers' online bio page indicates, "National Advisory Committee, Robert Wood Johnson Foundation Smoking Cessation Leadership Center, 2003-present."
see comments sharing old non-functional links
John R. Polito is solely responsible for the content of this article. Given its length and detail, the author acknowledges the possibility of factual errors. Any error will be immediately corrected upon receipt of credible authority in support of the writer's contention. E-mail comments to john@whyquit.com

