Nicotine gum, patch & lozenge a fraud, sham and hoax
Approved quitting products were sold as science, reassurance, and hope. But before they were truth, they were products. Before they were recovery, they were profits.

I wish it were true, that approved stop-smoking products were highly effective and that using them genuinely doubled a smoker’s odds of success. I wish I had broken free during my very first Nicorette nicotine gum attempt in about 1986. Instead, my spirit was left shattered.
And it wasn’t just me. By any honest measure, over-the-counter nicotine replacement therapy (NRT) products — the nicotine gum, patch, and lozenge — have not performed in the real world as their marketing taught smokers to expect.
I chewed my brains out. Still, when the chewing and parking cycles ended, the wanting for more nicotine remained. Soon, I was back to smoking as much as ever.
Was I somehow different? Had I just failed with the very best science had to offer? Was I hopeless?
Even in despair, I wasn’t finished trying.
Thinking the failure was mine, that I had somehow misused it, I wasted three more priceless periods of cessation confidence attempting to wean myself off nicotine by replacing it — once more with Nicorette gum and twice with the patch.
The cost ran deeper than wasted time, money, and confidence. Marketing that taught me nicotine was “medicine” and its use “therapy” also delayed critical learning. It obscured the real problem: the chemical my brain was dependent upon was still entering my body.
I had not only lost sight of the enemy. I had purchased and used it.
That matters. A smoker does not need to be taught that nicotine is missing. A smoker needs to learn how life feels once nicotine is gone — and stays gone.
Even so, year after year, the toll of smoke’s toxins was becoming more obvious and impossible to deny. I had to keep trying.
Although not yet known by name, repeated failed escape attempts would eventually force me to self-discover successful recovery’s only rule — the Law of Addiction (Chapter 2).
Free since May 15, 1999, I remain angry about decades of sham science displacing and suppressing truth for smokers desperate to quit. But anger is not this page’s purpose. Clarity is.
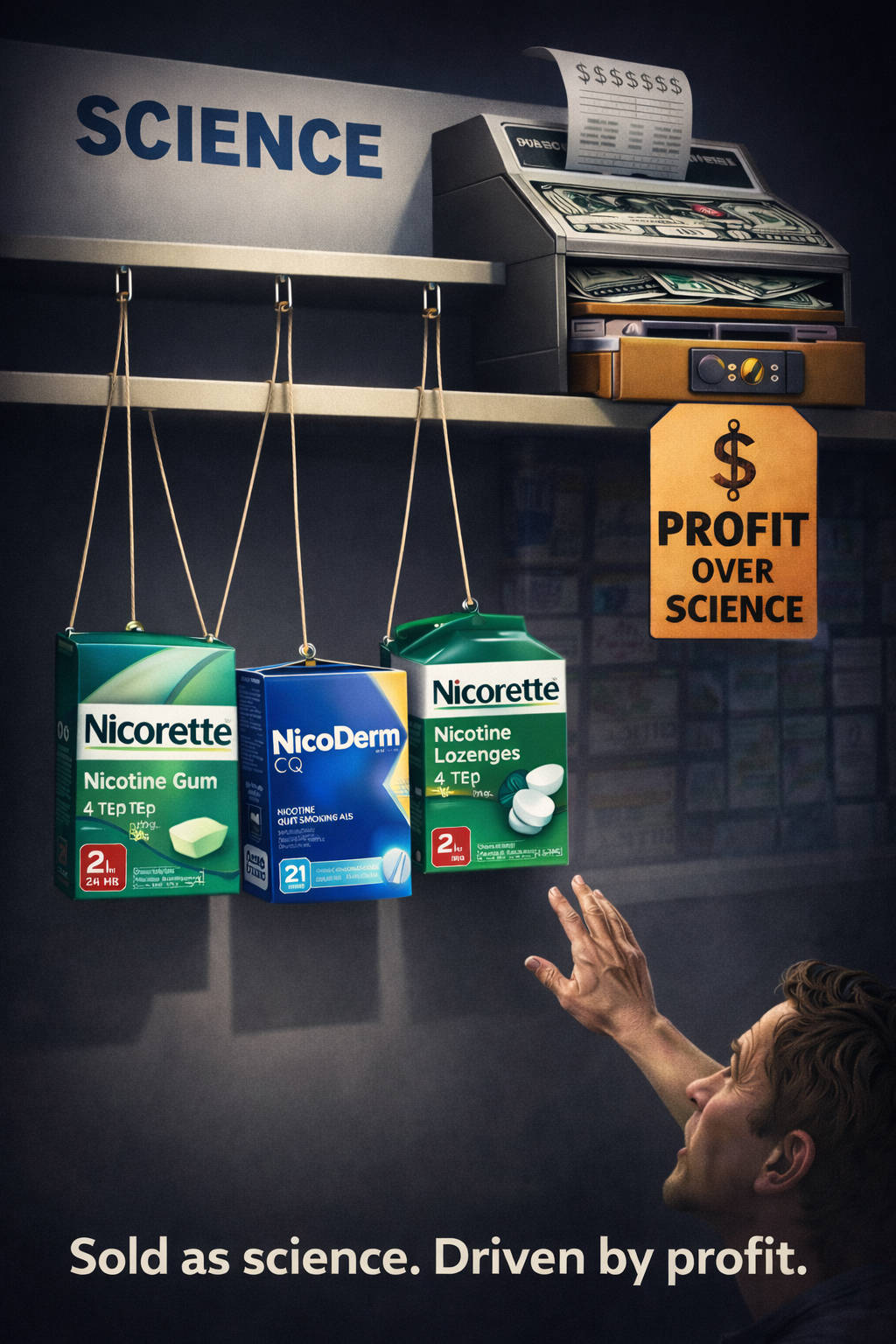
Knowledge is power. My hope is that what follows helps you instantly see the difference between a profitable quitting product and the truth about recovery.
Open lies and hidden truths
As I suggested in a 2012 letter published in the British Medical Journal, one of the most deadly health-product consumer frauds in history involves the study, marketing, and sale of over-the-counter replacement nicotine.[1]
The pharmaceutical industry and its allies have consistently fostered five impressions that collapse under honest inspection:
- That over-the-counter replacement nicotine has proven effective in real-world use;
- That most successful ex-smokers succeeded by using approved products;
- That few smokers are able to quit cold turkey;
- That placebo-controlled product studies were truly blind and trustworthy; and
- That product users soundly defeated cold turkey quitters in honest competition.
Understanding how smokers were sold those impressions also helps explain why cigarette companies now sell NRT too.[2]
It also clarifies the essence of addiction itself: dopamine-pathway wanting, urges, and desires make the neo-nicotine industry billions every year.
Falsehood #1: Nicorette helps you “chew away your cravings”
Let’s start with the most glaring consumer deception: the suggestion embedded in every nicotine gum advertisement most of us have ever seen — that nicotine gum has proven highly effective in helping smokers quit. Is it true?
In the United States, Nicorette was first approved for sale by the FDA on January 13, 1984. Decades later, and despite enormous marketing of newer products, nicotine gum remains one of the most promoted stop-smoking products in history.
A July 2013 Gallup Poll asked U.S. ex-smokers how they finally succeeded in quitting.[3] What percentage credited nicotine gum for their success?
- 41%
- 28%
- 13%
- 6%
- 1%
The correct answer was number five: 1%.
That single fact should stop every addicted reader cold. The most marketed stop-smoking product in history was credited by only 1% of successful ex-smokers.
Like an alcoholic toying with gradual stepped-down weaning schemes, it is nearly impossible for the brain to adjust to functioning without nicotine while nicotine continues to arrive. Especially when the replacement is reached for during the same situations in which nicotine was previously smoked, vaped, dipped, or chewed.
Falsehood #2: Most successful quitters use approved products
The same Gallup Poll found that all approved stop-smoking products combined — including Chantix — accounted for only a small fraction of successful quitting, just 8%, or about 1 in 12.[3]
Then, at long last, in 2020, even the U.S. Surgeon General openly acknowledged that “most smokers who quit successfully do so without medications or any type of formal assistance.”[4]
That is the truth most addicts were never told.
Approved products were sold as though they were the road to success, while the road most traveled by successful ex-smokers was left hidden in plain sight.
It is entirely normal for a craving addict who can satisfy urges within seconds of nicotine use to long for a quick quitting fix too. What is not normal is for the very institutions charged with protecting smokers to assist in presenting profit-driven mirages that prey upon that conditioned impulsiveness.
Smoking cessation became textbook proof that corporate greed can purchase the appearance of science, shape policy, and normalize group-think.
For example, the CDC’s SmokeFree.gov website devoted tabs to branded or approved products such as Nicorette, Nicoderm CQ, Nicotrol, Zyban, and Chantix.[5]
Which method isn't mentioned? Cold turkey — the path producing the most ex-smokers year after year and generates zero product profit.
Falsehood #3: Few are able to quit cold turkey
Truth is, there is a giant yet silent elephant in the room — a method that never advertises.
Reality is, as evidenced by the same Gallup Poll[3] and by independent population-level cessation studies,[6] each year more nicotine addicts arrest their dependence by going cold turkey than by all other methods combined.
How big is that elephant? Up to three-quarters who finally arrest their chemical dependence do so on their own, without using any product or procedure.[7]
According to the CDC, the United States had roughly 40 million adult smokers in 2014.[8] If about 6% quit successfully each year,[9] that means millions are still breaking free annually — and if 75% succeed without products, then unassisted quitting is not a fringe method. It is the main event.
So how is it possible that such a massive truth — how most people actually succeed — remained hidden, while approved products prevailed in clinical headlines yet got clobbered in real-world use?[10]
Because products are easier to sell than confidence. A chemical can be packaged. Freedom cannot.
Ask yourself: is your dream to keep feeding your dependence until the day you die, or to arrest it?
If you are less than 72 hours away from clearing nicotine from your body and moving beyond peak withdrawal, what sense does it make to pay money to extend nicotine use for weeks or months, or to risk becoming the cure’s permanent customer?
Cold turkey is fast, free, effective, and smart
You have been lied to for so long that skepticism here is normal. The flip side of the lie that NRT is highly effective is the lie that cold turkey is not.
Again, in 2020, the U.S. Surgeon General at long last acknowledged that numerous population surveys indicate “that cold-turkey quitters do as well or better than those who use over-the-counter NRTs.”[4]
Despite the vast majority of successful ex-users having abruptly ended nicotine use without products or procedures, industry influence continues to suggest that few succeed that way, that you would need to be some kind of super-hero to do so.
That is why quit-method surveys are relatively rare. The medicinization and commercialization of cessation depend on your not knowing the truth.
Since 1984, billions have been spent teaching smokers to distrust their natural recovery instincts.
Reflect on what decades of “double your chances” messaging have done to confidence. Imagine trying to navigate early withdrawal while being told you are probably failing unless you buy and use a product.
The damage is hard to overstate. That message has eroded confidence, encouraged relapse, and cost freedom-seeking dreamers both opportunities and years of life.
Never in history has a greater array of approved products promised to double success. And never have the social, legal, and financial pressures to stop smoking been greater.
Do you really think you will stop once the price rises a few more dollars, euros, or pounds? So did millions of smokers in countries already paying some of the world’s highest cigarette prices.[11]
Reflect too on the millions of addicts willing to risk frightening Chantix side effects because marketing taught them to distrust the fastest, cheapest, and most widely successful path to freedom.
The tragedy is that the number two quitting method behind cold turkey is not the gum, patch, or Chantix. It's smoking yourself to death.
In the U.S., millions of smoking-related deaths occurred even during the years when pharmaceutical cessation advertising and promotion were everywhere.[12]
And that was while billions were spent trying to convince smokers to buy replacement nicotine, risk Chantix or Champix,[13] or chase any number of other paid “solutions.”[14]
A 2006 Australian study analyzing the smoking patients of 1,000 family practice physicians found that 88% of successful ex-smokers succeeded by going cold turkey and that those going cold were twice as likely to succeed as those using the patch, gum, inhaler, or Zyban.[15]
Still, nicotine addicts make easy prey. Why? Because thousands of daily urges were once satisfied within seconds. Because old satisfaction memories teach the prisoner to dream of a fast, painless, surefire escape.
And we dream in an arena where almost any quitting scheme — even a ridiculous one — can generate testimonials by some percentage of users at six months.[16]
That matters because the natural six-month success rate per quit attempt among those stopping entirely on their own is not zero. A product does not need to be miraculous to generate testimonials. It only needs to be present while some people succeed.
Do not fret. That is why this book exists: to shine light on darkness and to emphasize that it is IMPOSSIBLE to fail so long as all nicotine remains on the outside.
To help explain cessation fraud, pretend we invent a new magic stop-smoking product called “Billy Bob’s Lima Bean Butter.”
Unless our product somehow undercuts natural cessation, some percentage of users should still succeed and remain smoke-free at six months.[17]
The sad part is that nearly all who succeed while using Billy Bob’s Lima Bean Butter would deeply believe the butter was responsible. Even after hearing the truth, many still would.
And we can improve our sham further by pairing the butter with recovery interventions that have their own independent effectiveness.
For example, we could combine butter use with counseling, coping-skills development, or behavioral support, all of which have been shown to improve outcomes.[18]
Now our product gets the credit while the true source of improvement is blurred. That inflation formula has shadowed countless clinical studies of NRT, Zyban, and Chantix/Champix.
For example, Pfizer’s original Chantix studies included record-setting numbers of counseling contacts, while subsequent marketing continued to award full credit to Chantix itself.[19]
While approved products defeat the expectations of placebo users inside highly supported clinical trials, their real-world performance has repeatedly disappointed.
California,[20] Massachusetts,[21] Minnesota,[22] Quebec,[23] London,[24] Western Maryland,[25] Nottingham,[26] Australia,[27] the National Cancer Institute,[28] England’s Stop Smoking Services,[29] TUS-CPS data,[30] English households,[31] the U.S. KnowledgePanel,[32] PATH data,[33] TUS-CPS 2010–2011,[34] and later PATH data again[35] all point in the same uncomfortable direction: over-the-counter approved products have not delivered the real-world superiority their marketing implied.
Despite quit-method surveys being cheap and easy to conduct, they are relatively rare compared to the mountain of clinical efficacy trials. That is not an accident.
Once the absence of favorable real-world evidence became glaring, something had to be done.
So how did pharma and its most trusted researchers respond? They began writing and conducting their own surveys.
Those surveys often overemphasized approved products through repeated questioning while avoiding direct mention of cold turkey. Unassisted quitters were pushed into categories such as “other” or “none,” or their method was ignored entirely.[36] [37]
It also gave financially conflicted researchers control over raw data, exclusion criteria, and statistical adjustments before publication.[38]
Meanwhile, successful ex-users had no reason to lie about how they finally quit, yet unless a survey aligned with industry-friendly methodology it was often dismissed as inferior.
Imagine telling ex-smokers they cannot be trusted to remember the last method that finally worked, unless the survey itself was built inside pharma’s worldview.
Pharma and its defenders also argue that many go cold turkey only because they lack insurance coverage or cannot afford NRT.[4]
This despite the fact that continuing to smoke is often more expensive than many approved products, and despite the reality that what smokers want most is not another customer relationship with nicotine, but freedom.
They also argue that cold turkey only appears stronger in surveys because “heavier” smokers choose NRT. But selection does not prove product effectiveness. And it certainly does not justify hiding how most successful quitters actually succeeded.
The larger selection bias is the one almost never acknowledged: more than 200 NRT clinical trials recruited people by dangling free NRT or “medication” in front of smokers who were already hoping to reduce withdrawal by using a product.
Those dreaming of going cold were not there.
Which is more deadly: telling smokers the truth about how most succeed, or lying to them about “their chances” unless they buy a product?
Real blame belongs with greed that turned nicotine into “medicine,” dependency into “therapy,” and recovery into a market.
Frankly, what cannot be trusted is any clinical trial claim whose persuasive force depends on comparing an active product to placebo and then presenting the result as though it settled the real-world question.
References
- 1. Polito, JR, Are those who quit smoking paying with their lives because of NRT's failure? British Medical Journal, February 7, 2012, Page 344, e866.
- 2. Michael Feldberbaum, Tobacco Giant Reynolds Wants To Be A Major Nicotine Gum Company, AP, September 4, 2014 (story relates how U.S. sales of RJ Reynolds Zonnic brand nicotine gum are going nationwide).
- 3. Gallup. Most U.S. smokers want to quit, have tried multiple times. July 31, 2013.
- 4. U.S. Department of Health and Human Services. Smoking Cessation. A Report of the Surgeon General, January 2020, Atlanta, GA.
- 5. SmokeFree.gov, Find a Quit Method That Works For You, reviewed March 29, 2016.
- 6. Cancer Council Australia, Most Australians still quit smoking unassisted, Oct. 8, 2010; also see Chapman S, MacKenzie R. The Global Research Neglect of Unassisted Smoking Cessation: Causes and Consequences. PLoS Med. 2010;7(2):e1000216.
- 7. Chapman S, MacKenzie R. The global research neglect of unassisted smoking cessation: causes and consequences. PLoS Med. 2010;7(2):e1000216; also see Doran CM, et al. Smoking status of Australian general practice patients and their attempts to quit. Addictive Behaviors. 2006;31(5):758-766.
- 8. CDC, Smoking & Tobacco Use - Fast Facts.
- 9. CDC, Morbidity and Mortality Weekly Report (MMWR), Quitting Smoking Among Adults - United States, 2001-2010.
- 10. Doran CM, et al. Smoking status of Australian general practice patients and their attempts to quit. Addictive Behaviors. 2006;31(5):758-766; also see Polito, JR, Replacement Nicotine's Killing Fields, WhyQuit.com, February 11, 2012.
- 11. InsiderMonkey.com, 10 Most Expensive Countries to Buy Cigarettes in the World, July 24, 2015.
- 12. Compare: CDC, Cigarette Smoking Among Adults - United States, 2007, November 28, 2008, MMWR Vol57, No. 45 with CDC, Vital Signs: Current Cigarette Smoking Among Adults Aged 18 Years - United States, 2005-2010.
- 13. Polito, JR, "Will Chantix really help me quit smoking?" WhyQuit.com, August 25, 2006.
- 14. Polito, JR, Smoke Remedy infomercial: a fraud, hoax and scam? WhyQuit.com, January 10, 2011.
- 15. Doran CM, et al. Smoking status of Australian general practice patients and their attempts to quit. Addictive Behaviors. 2006;31(5):758-766; also see Polito, JR, Cold Turkey Twice as Effective as NRT or Zyban, WhyQuit.com, May 19, 2006.
- 16. Polito, JR, Does the Over-the-counter Nicotine Patch Really Double Your Chances of Quitting? WhyQuit.com, April 8, 2002.
- 17. Hughes JR, Shiffman S, et al. A meta-analysis of the efficacy of over-the-counter nicotine replacement. Tobacco Control. 2003;12:21-27.
- 18. Polito, JR, FDA Chantix Handling Betrayed Public Health, WhyQuit.com, February 29, 2008.
- 19. Polito, JR, Is a 14% Chantix success rate worth risking death? WhyQuit.com, June 14, 2011; also see Gonzales D, et al. Varenicline vs sustained-release bupropion and placebo for smoking cessation: a randomized controlled trial. JAMA. 2006;296(1):47-55.
- 20. Pierce JP, et al. Impact of over-the-counter sales on effectiveness of pharmaceutical aids for smoking cessation. JAMA. 2002;288:1260-1264.
- 21. Alpert HR, Connolly GN, Biener L. A prospective cohort study challenging the effectiveness of population-based medical intervention for smoking cessation. Tobacco Control. 2012.
- 22. Boyle RG, et al. Does insurance coverage for drug therapy affect smoking cessation? Health Affairs. 2002;21(6):162-168.
- 23. Gomez-Zamudio M, et al. Role of pharmacological aids and social supports in smoking cessation associated with Quebec's 2000 Quit and Win campaign. Preventive Medicine. 2004;38(5):662-667.
- 24. SmokeFree London, Tobacco In London, Facts and Issues, November 26, 2003.
- 25. Alberg AJ, et al. Nicotine replacement therapy use among a cohort of smokers. Journal of Addictive Diseases. 2005;24(1):101-113.
- 26. Ferguson J, et al. The English smoking treatment services: one-year outcomes. Addiction. 2005;100 Suppl 2:59-69.
- 27. Doran CM, et al. Smoking status of Australian general practice patients and their attempts to quit. Addictive Behaviors. 2006;31(5):758-766.
- 28. Hartman AM. What does US national population survey data reveal about effectiveness of nicotine replacement therapy on smoking cessation? World Conference on Tobacco or Health, July 2006; also see Pierce JP, et al. Quitlines and nicotine replacement for smoking cessation: do we need to change policy? Annual Review of Public Health. 2012;33:341-356.
- 29. UK NHS, Statistics on NHS Stop Smoking Services in England, April to December 2007.
- 30. Pierce JP, et al. Quitlines and nicotine replacement for smoking cessation: do we need to change policy? Annual Review of Public Health. 2012;33:341-356.
- 31. Kotz D, et al. Prospective cohort study of the effectiveness of smoking cessation treatments used in the "real world." Mayo Clinic Proceedings. 2014;89(10):1360-1367.
- 32. Weaver SR, et al. Are electronic nicotine delivery systems helping cigarette smokers quit? Evidence from a prospective cohort study of U.S. adult smokers, 2015-2016. PLoS ONE. 2018;13(7):e0198047.
- 33. Benmarhnia T, et al. Can e-cigarettes and pharmaceutical aids increase smoking cessation and reduce cigarette consumption? American Journal of Epidemiology. 2018;187(11):2397-2404.
- 34. Leas EC, et al. Effectiveness of pharmaceutical smoking cessation aids in a nationally representative cohort of American smokers. Journal of the National Cancer Institute. 2018;110(6):581-587.
- 35. Pierce JP, et al. Role of e-cigarettes and pharmacotherapy during attempts to quit cigarette smoking: The PATH Study 2013-16. PLOS One. 2020.
- 36. USDHHS, Population Assessment of Tobacco and Health (PATH) Study, Wave 3: Adult Questionnaire Data, updated April 19, 2018.
- 37. West R, Smokers Toolkit February 2010 Wave 40 - Final Questionnaire, Smoking in England, 2010.
- 38. Polito JR, Pharma bias destroys ITC quit smoking medication study, August 20, 2012, reviewing Kasza KA, et al. Effectiveness of stop-smoking medications: findings from the ITC Four Country Survey. Addiction. 2013;108(1):193-202.
- 39. Walmart.com, search Nicotine Replacement Therapy, used per unit prices for Equate NRT product line. Date searched August 23, 2020.